Diatomic element definition chemistry9/16/2023  Atoms will react to get in the most stable state possible. The octet rule refers to the tendency of atoms to prefer to have eight electrons in the outermost shell. Let us discuss a bit about what this rule is. It is shown in letters. Now, why are the atoms more stable when they are in pairs? It is because they obey the octet rule. The horizontal line represents a bond between the pair of atoms. In the image below you can see the dot structures for F2, Cl2, Br2, I2, and H2. In this type of element, there is a pair of atoms with a chemical bond. The diatomic elements are elements that appear as molecules. And this is the same for all the seven elements. When they put chlorine in the pool, it’s Cl2 (not Cl). Similarly, even though the periodic table only lists H, a canister of hydrogen gas would contain H2. Though it shows N on the periodic table, in the atmosphere it is N2. There is also a lot of nitrogen in the atmosphere. However, based on the context, it’s usually pretty obvious which is which. But if they are talking about an actual substance they probably mean O2. If they are talking about atoms or the periodic table, they probably mean O. 
Still, most of the time people just say “oxygen”. So, O atoms are just O, and oxygen gas means O2. But the atoms live in pairs to make O2 oxygen gas. The logic here is that O is an oxygen atom. However, the oxygen present in the atmosphere is shown by the letter O2. On the periodic table, the element oxygen is shown by the letter O. Even if the air has O2, you will never find O atoms floating around in the environment. However, oxygen is also the gas that helps us to stay alive. Oxygen is the letter O on the periodic table. The naming system for these elements is somewhat lacking. These weights were a necessary factor in the discovery of the periodic law by Dmitri Mendeleev and Lothar Meyer. Also, because of apparent exceptions to Avogadro’s law were not explained until later.Ĭannizzaro revived Avogadro’s ideas and used them to create a database of atomic weights. Partly because it was thought that atoms of one element would have no chemical affinity for atoms of the same element. However, these results were mostly ignored until 1860. And also, the assumption of diatomic elemental molecules. Based on these stands the now called Avogadro’s law. By 1811, Amedeo Avogadro had successfully arrived at the proper chemical analysis of water. In 1805, Gay-Lussac and von Humboldt demonstrated that water is two volumes of hydrogen and one volume of oxygen. As a result, there was a misunderstanding regarding atomic weights and molecular formulas. It meant that the atomic weight of oxygen was eight times that of hydrogen. For example, Dalton assumed that water had the formula HO. He also assumed that atoms in compounds have the simplest atomic ratios among themselves. He had assumed that all elements had only one atom. John Dalton came up with the initial atomic hypothesis. It is because we find some of the most common elements, as diatomic (e.g. In the 19th century, these elements were very much important in the development of the concepts of element, atom, and molecule. As a result, H2 is the most abundant among them. However, in the Earth’s atmosphere, the natural abundance of hydrogen (H2) is on the order of parts per million. These are nitrogen (78 %) and oxygen (29 %). The Earth’s atmosphere consists of two of these molecules. We also find them in the laboratory, and in interstellar space. Hundreds of compounds have been discovered in the Earth’s environment. This is a perfect example of this type of element. Therefore, two O atoms make bonds and create O2. These atoms are so unstable that they cannot exist alone. 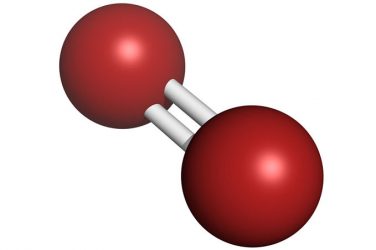
Oxygen exists as O2 because the O atoms must live in pairs. You will never find lone O atoms floating around the atmosphere. There has to be a bond of at least two atoms present. So, when we say a molecule is diatomic, we mean that atom cannot exist by itself. Take a look at the prefix ‘di-‘ in the word. You will find the answer in the word itself. So, this oxygen, O2, and nitrogen, N2, are part of this element group. You will find that the atmosphere is composed of almost 78% nitrogen and 21% of oxygen. Now, take a look at the distribution of these gases in the atmosphere. These are elements like nitrogen, oxygen, a little amount of argon, and some other gases. When we study the structure of the earth, we see there are major components that make up the earth’s atmosphere. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
